Peer-Reviewed Publications
For a complete up-to-date list of publications please go to my ORCID or Google Scholar pages.
2018 Lopex-Baez, JC., Zeng, Z., Rybski, W., Huitema, LFA., Dale, RM., Kawakami, K., Englert, C., Schulte-Merker, S., Hastie, ND., and
Patton, EE. Wilms Tumor 1b defines a wound-specific sheath cell subpopulation associated with notochord repair. eLife 7:e30657 https://doi.org/10.7554/eLife.30657
2017 Jones, RN, Erhard, SA, Malham, MR, Gen, AY, Sullivan, K, Olsen, KW, and Dale, RM. Expression and characterization of the
zebrafish orthologue of the human FOLR1 gene during embryogenesis. Gene Expression Patterns, 25-26:159-166
https://doi.org/10.1016/j.gep.2017.08.002
2015 Sisson, BE, Dale, RM, Mui, SR, Topczewska, JM, and, Topczewski, J. A role of glypican4 and wnt5b in chondrocyte stacking
underlying craniofacial cartilage morphogenesis. Mechanisms of Development 138:279-290
https://doi.org/10.1016/j.mod.2015.10.001
2015 Askary, A., Mork, L., Paul, S., He, X., Izuhara, A. K., Gopalakrishnan, S., Ichida, J. K., McMahon, A. P., Dabizljevic, S., Dale, RM., Mariani, F. V., Crump, J. G. Iroquois Proteins Promote Skeletal Joint Formation by Maintaining Chondrocytes in an Immature
State. Developmental Cell. 35:358-365 https://doi.org/10.1016/j.devcel.2015.10.004
2014 Gray, R. S., Wilm, T.P., Smith, J., Bagnat, M., Dale, RM, Topczewski, J., Johnson, S. L., Solnica-Krezel, L. Loss of col8a1a function
during zebrafish embryogenesis results in congenital vertebral malformations. Developmental Biology. 386:72-85
https://doi.org/10.1016/j.ydbio.2013.11.028
2014 Antoine, T. E., Jones, K. S., Dale, RM., Shukla, D., Tiwari, V. Zebrafish: Modeling for Herpes Simplex Virus Infections. Zebrafish
11:17-25. https://doi.org/10.1089/zeb.2013.0920
2011 Topczewski, J, Dale, RM, and Sisson, BE. PCP signaling in Craniofacial Development. Organogenesis, 7:255-59.
http://dx.doi.org/10.4161/org.7.4.18797
2011 Dale, RM, and Topczewski, J, Identification of an evolutionarily conserved regulatory element of the zebrafish col2a1a
gene. Developmental Biology, Vol. 357:518-531. http://doi.org/10.1016/j.ydbio.2011.06.020
2010 Demonbreun, AR, Lapidos, KA, Heretis, K, Levin, S, Pytel, P, Dale, RM, Svensson, EC, McNally, EM. Myoferlin regulation by NFAT
in muscle injury, regeneration, and repair. Journal of Cell Science, 123:2413-22. https://doi.org/10.1242/jcs.065375
2009 Dale, RM, Sisson, BE, and Topczewski, J. The emerging role of Wnt/PCP signaling in organ formation. Zebrafish, 6:9-14.
https://doi.org/10.1089/zeb.2008.0563
2007 Dale, RM, Remo, BF, and Svensson, EC. An alternative transcript of the FOG-2 gene encodes a FOG-2 isoform lacking the FOG
repression motif. Biochemical and Biophysical Research Communications, 357: 683-687.
https://doi.org/10.1016/j.bbrc.2007.04.008
2018 Lopex-Baez, JC., Zeng, Z., Rybski, W., Huitema, LFA., Dale, RM., Kawakami, K., Englert, C., Schulte-Merker, S., Hastie, ND., and
Patton, EE. Wilms Tumor 1b defines a wound-specific sheath cell subpopulation associated with notochord repair. eLife 7:e30657 https://doi.org/10.7554/eLife.30657
2017 Jones, RN, Erhard, SA, Malham, MR, Gen, AY, Sullivan, K, Olsen, KW, and Dale, RM. Expression and characterization of the
zebrafish orthologue of the human FOLR1 gene during embryogenesis. Gene Expression Patterns, 25-26:159-166
https://doi.org/10.1016/j.gep.2017.08.002
2015 Sisson, BE, Dale, RM, Mui, SR, Topczewska, JM, and, Topczewski, J. A role of glypican4 and wnt5b in chondrocyte stacking
underlying craniofacial cartilage morphogenesis. Mechanisms of Development 138:279-290
https://doi.org/10.1016/j.mod.2015.10.001
2015 Askary, A., Mork, L., Paul, S., He, X., Izuhara, A. K., Gopalakrishnan, S., Ichida, J. K., McMahon, A. P., Dabizljevic, S., Dale, RM., Mariani, F. V., Crump, J. G. Iroquois Proteins Promote Skeletal Joint Formation by Maintaining Chondrocytes in an Immature
State. Developmental Cell. 35:358-365 https://doi.org/10.1016/j.devcel.2015.10.004
2014 Gray, R. S., Wilm, T.P., Smith, J., Bagnat, M., Dale, RM, Topczewski, J., Johnson, S. L., Solnica-Krezel, L. Loss of col8a1a function
during zebrafish embryogenesis results in congenital vertebral malformations. Developmental Biology. 386:72-85
https://doi.org/10.1016/j.ydbio.2013.11.028
2014 Antoine, T. E., Jones, K. S., Dale, RM., Shukla, D., Tiwari, V. Zebrafish: Modeling for Herpes Simplex Virus Infections. Zebrafish
11:17-25. https://doi.org/10.1089/zeb.2013.0920
2011 Topczewski, J, Dale, RM, and Sisson, BE. PCP signaling in Craniofacial Development. Organogenesis, 7:255-59.
http://dx.doi.org/10.4161/org.7.4.18797
2011 Dale, RM, and Topczewski, J, Identification of an evolutionarily conserved regulatory element of the zebrafish col2a1a
gene. Developmental Biology, Vol. 357:518-531. http://doi.org/10.1016/j.ydbio.2011.06.020
2010 Demonbreun, AR, Lapidos, KA, Heretis, K, Levin, S, Pytel, P, Dale, RM, Svensson, EC, McNally, EM. Myoferlin regulation by NFAT
in muscle injury, regeneration, and repair. Journal of Cell Science, 123:2413-22. https://doi.org/10.1242/jcs.065375
2009 Dale, RM, Sisson, BE, and Topczewski, J. The emerging role of Wnt/PCP signaling in organ formation. Zebrafish, 6:9-14.
https://doi.org/10.1089/zeb.2008.0563
2007 Dale, RM, Remo, BF, and Svensson, EC. An alternative transcript of the FOG-2 gene encodes a FOG-2 isoform lacking the FOG
repression motif. Biochemical and Biophysical Research Communications, 357: 683-687.
https://doi.org/10.1016/j.bbrc.2007.04.008
Book Chapters
2008 Svensson, EC, Wilk, J, Dale, RM, and Modrell, M. “The role of the transcriptional co-repressor FOG-2 in cardiac development” in, Cardiovascular Development and Congenital Malformations Artman, M., Benson, D.W., Srivastava, D., and Nakazawa, M. (eds), p125-127.
COVER ARTWORK
Identification of an evolutionarily conserve regulatory element of the zebrafish col2a1a gene

Zebrafish (Danio rerio) is an excellent model organism for the study of vertebrate development including skeletogenesis. Studies of mammalian cartilage formation were greatly advanced through the use of a cartilage specific regulatory element of the Collagen type II alpha 1 (Col2a1) gene. In an effort to isolate such an element in zebrafish, we compared the expression of two col2a1 homologues and found that expression of col2a1b, a previously uncharacterized zebrafish homologue, only partially overlaps with col2a1a. We focused our analysis on col2a1a, as it is expressed in both the stacked chondrocytes and the perichondrium. By comparing the genomic sequence surrounding the predicted transcriptional start site of col2a1a among several species of teleosts we identified a small highly conserved sequence (R2) located 1.7 kb upstream of the presumptive transcriptional initiation site. Interestingly, neither the sequence nor location of this element is conserved between teleost and mammalian Col2a1. We generated transient and stable transgenic lines with just the R2 element or the entire 1.7 kb fragment 5′ of the transcriptional initiation site. The identified regulatory elements enable the tracking of cellular development in various tissues by driving robust reporter expression in craniofacial cartilage, ear, notochord, floor plate, hypochord and fins in a pattern similar to the expression of endogenous col2a1a. Using a reporter gene driven by the R2 regulatory element, we analyzed the morphogenesis of the notochord sheath cells as they withdraw from the stack of initially uniform cells and encase the inflating vacuolated notochord cells. Finally, we show that like endogenous col2a1a, craniofacial expression of these reporter constructs depends on Sox9a transcription factor activity. At the same time, notochord expression is maintained after Sox9a knockdown, suggesting that other factors can activate expression through the identified regulatory element in this tissue.
IROQUOIS PROTEINS PROMOTE SKELETAL JOINT FORMATION BY MAINTAINING CHONDROCYTES IN AN IMMATURE STATE.
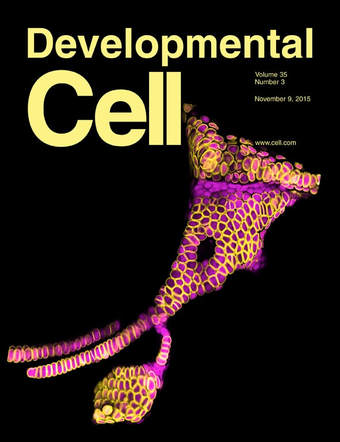
An early event in skeletal joint development is the specification of articular chondrocytes at the joint surface. Articular chondrocytes are distinct in producing lower levels of cartilage matrix and not being replaced by bone, yet how they acquire these properties remains poorly understood. Here, we show that two members of the Iroquois transcriptional repressor family, Irx7 and Irx5a, function to block chondrocyte maturation at the developing hyoid joint of zebrafish. These Irx factors suppress the production of cartilage matrix at the joint in part by preventing the activation of a col2a1a enhancer by Sox9a. Further, both zebrafish Irx7 and mouse IRX1 are able to repress cartilage matrix production in a murine chondrogenic cell line. Iroquois proteins may therefore have a conserved role in keeping chondrocytes in an immature state, with the lower levels of cartilage matrix produced by these immature cells contributing to joint flexibility.
